People Are Dying Because We Can't Place 10 Stickers on a Chest Correctly
Imagine this: A 52-year-old man collapses at a family barbecue. His wife calls 911. Paramedics arrive in seven minutes. They place electrodes on his chest and run a 12-lead ECG. The tracing looks abnormal — but not in the way you'd expect. The ST segments are elevated in leads V1 and V2, triggering a STEMI alert. The cath lab is activated. An interventional cardiologist is pulled out of bed at 2 a.m. A team of nurses and techs race to the hospital. Sterile equipment is opened. Anticoagulants are administered.
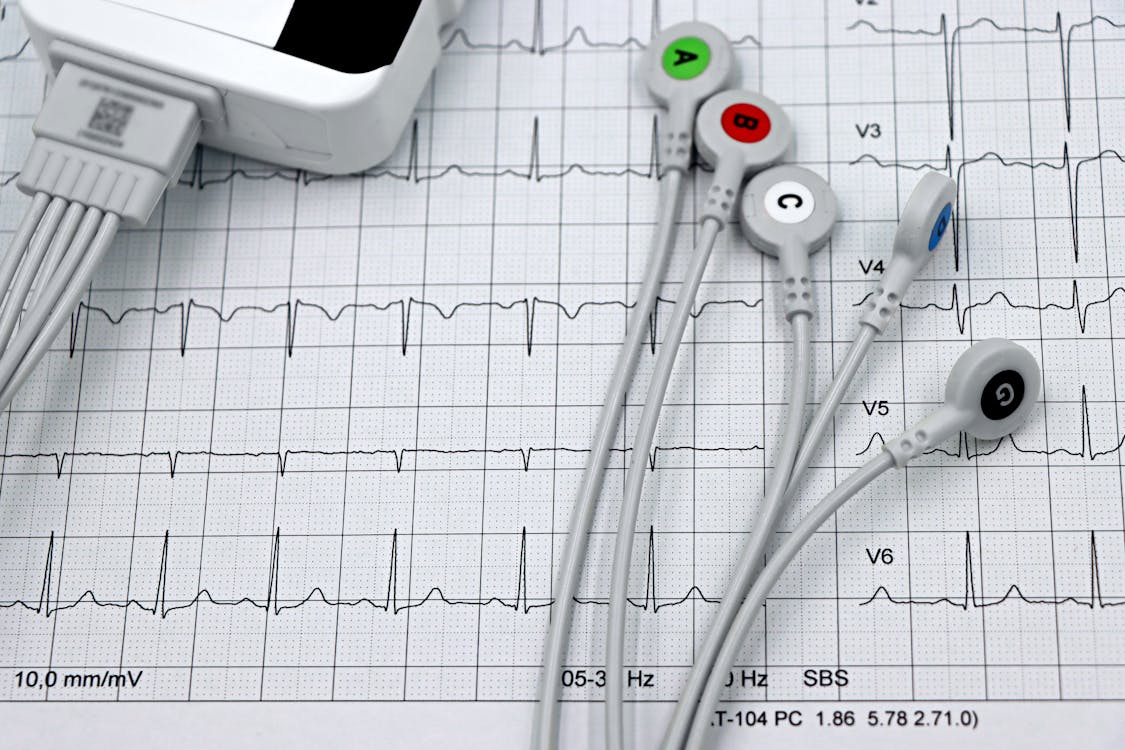
The Alarming Failure Rate of ECG Electrode Placement — and the Trail of Preventable Deaths, Misdiagnoses, and Billions in Wasted Revenue It Leaves Behind
Imagine this: A 52-year-old man collapses at a family barbecue. His wife calls 911. Paramedics arrive in seven minutes. They place electrodes on his chest and run a 12-lead ECG. The tracing looks abnormal — but not in the way you’d expect. The ST segments are elevated in leads V1 and V2, triggering a STEMI alert. The cath lab is activated. An interventional cardiologist is pulled out of bed at 2 a.m. A team of nurses and techs race to the hospital. Sterile equipment is opened. Anticoagulants are administered.
But this man is not having a heart attack.
The electrodes were placed two intercostal spaces too high. The “STEMI” was an artifact. A ghost. And now, thousands of dollars have been burned, a cath lab team is exhausted from another false alarm, and somewhere else in the city, a real STEMI patient is waiting for a team that’s already deployed.
Now imagine the opposite: A 47-year-old woman with crushing chest pain receives a prehospital ECG. The electrodes are misplaced just slightly — too lateral, too low. Her active anterior wall MI is obscured. The tracing looks unremarkable. She is transported to a community hospital without PCI capability. By the time the real diagnosis is made, irreversible myocardial damage has occurred.
Both of these scenarios happen every single day across the United States. They are not rare. They are not edge cases. They are the predictable, documented, and devastating consequences of a systemic failure that has been hiding in plain sight for over 70 years.
The Numbers That Should Keep Every Clinician Awake at Night
The data is not ambiguous. It is damning.
94.2%
of paramedics misplace at least one chest electrode. Only 5.8% achieved correct placement of all six precordial leads. (Gregory et al., British Paramedic Journal, 2021)
64%
of nurses misplace ECG leads. Cardiologists correctly place V1 and V2 less than 20% of the time. (Hadjiantoni et al., Cardiology and Cardiovascular Medicine, 2021)
Up to 72%
of inappropriate cath lab activations are caused by ECG misinterpretation — misinterpretation that frequently begins with misplaced electrodes. (Degheim et al., JRSM Cardiovascular Disease, 2019)
36%
of STEMI cath lab activations from EDs were false positives in a two-center study at Brigham and Women’s Hospital / Harvard. (McCabe et al., Archives of Internal Medicine, 2012)
7.5%
relative increase in one-year mortality risk for every 30 minutes of treatment delay in STEMI patients undergoing primary PCI (adjusted RR 1.075 per 30 min). (De Luca et al., Circulation — AHA Journals, 2004)
Read that last number again. Every 30 minutes of delay is associated with a 7.5% relative increase in the risk of one-year mortality. A misplaced electrode that masks a real STEMI or triggers a false alarm that diverts resources is not just an inconvenience. It is a contributor to mortality. Published in Circulation, the flagship journal of the American Heart Association, this finding underpins the urgency behind every AHA and ACC guideline on STEMI management.
The Anatomy of a Failure That Starts With 2 Centimeters
ECG morphology changes become clinically significant when electrodes are displaced by as little as 2 centimeters from the correct position (Kania et al., 2014). Two centimeters. The width of a thumb.
When V1 and V2 — the two most critical precordial leads for detecting anterior and septal wall cardiac events — are placed too high (in the 2nd or 3rd intercostal space instead of the 4th), the resulting ECG can generate erroneous patterns including ST-segment elevation, incomplete right bundle branch block, anterior T-wave inversion, and septal Q waves (Walsh, American Journal of Emergency Medicine, 2018). These artifacts can mimic a STEMI with alarming fidelity.
Conversely, when electrodes are displaced inferiorly or laterally, a genuine ST-elevation pattern can be flattened or eliminated entirely. The heart attack is there. The ECG just can’t see it.
⚠ WARNING: Researchers have also documented that limb leads are frequently placed on the torso instead of the extremities, particularly in prehospital settings. This practice can mask inferior infarcts while fabricating false-positive lateral infarcts — sending care teams down a completely wrong clinical pathway. (Walsh, 2018; EMS1, 2025)
A 2024 study in the International Journal of Paramedicine found that only 41.6% of both EMS and clinical personnel achieved “acceptable” electrode placement, with 34.2% placing three or fewer electrodes acceptably (Clopton & Hyrkäs, 2024). The problem is not limited to any one discipline. It spans paramedics, EMTs, nurses, ED physicians, and even cardiologists. Medani et al. (2018) found that only 10% of participants — across all clinical roles — placed all leads correctly.
This is not a human error problem. This is a systems design failure. The tools we use to acquire the most important cardiac diagnostic test have not meaningfully changed since the 1950s.
False Cath Lab Activations: A Hemorrhage of Time, Money, and Trust
When the AHA and ACC issued guidelines recommending cath lab activation within 10 minutes of the first ECG suggesting STEMI, the intention was lifesaving. And it has saved lives. But it also opened the floodgates to a crisis no one anticipated.
Before 2013, inappropriate cath lab activations accounted for fewer than 10% of all activations. Since then, false activation rates have surged to between 15% and 40%, with some institutions reporting rates as high as 40% of all emergent activations not requiring emergent cardiac intervention (Degheim et al., 2019; University of Michigan, AJMC, 2013).
Each false activation triggers an expensive cascade:
| Resource Consumed | Impact |
|---|---|
| Interventional cardiologist | Called in (often off-hours, middle of night) |
| Cath lab nursing & tech team | Mobilized, suited, prepped — for nothing |
| Sterile equipment & supplies | Opened and wasted (cannot be re-sterilized) |
| Medications administered | Anticoagulants, antiplatelets given to a non-STEMI patient |
| Per-activation cost estimate | $350–$865+ per false activation (Cardiovascular Diagnosis & Therapy) |
| Cath lab operational cost | ~$17,000 avg. procedural cost per case (MedAxiom) |
But the financial cost, as staggering as it is, may not even be the most dangerous consequence.
⚠ WARNING: A study published in the Journal of the American Heart Association found that repeated false cath lab activations contribute to staff burnout and — critically — a loss of urgency over time. When the team has been burned by false alarms repeatedly, their response to the real emergency becomes slower, less precise, and less focused. This is the “boy who cried wolf” effect in a setting where every minute of delay costs human life.
The concept of “time is muscle” in STEMI care is well established. De Luca et al., publishing in Circulation (AHA), demonstrated that the risk of one-year mortality increases by 7.5% for every 30 minutes of treatment delay in primary PCI (De Luca et al., 2004). A JACC-published analysis of mortality trends in STEMI estimated the broader impact at approximately 10% increased mortality risk per 60 minutes of delay. For STEMI patients in cardiogenic shock, the FITT-STEMI trial (n=12,675, published in the European Heart Journal, 2018) found that every 10 minutes of treatment delay resulted in 3.31 additional deaths per 100 PCI-treated patients — a mortality escalation roughly 10 times higher than in hemodynamically stable patients (0.34 per 100).
When a false alarm diverts the cath lab team, the next patient with a real STEMI may face a delay that is measured not in minutes, but in irreversible damage to heart muscle — and possibly their life.
The Other Side of the Coin: When Electrode Misplacement Hides a Heart Attack
While false activations waste resources, the inverse scenario is arguably even more terrifying: a true STEMI that is missed because misplaced electrodes produced a normal-looking tracing.
Research compiled by Newman-Toker et al. (2022) for the Agency for Healthcare Research and Quality identified approximately 10,000 potentially preventable harms related to missed acute myocardial infarction annually in the United States. Industry analysis estimates a 27.5% annual U.S. missed or delayed MI detection rate and approximately $2.4 billion in annual heart attack wrongful death exposure, derived from published misdiagnosis rates, malpractice win rates, and average indemnity payments (Newman-Toker et al., 2022; CBI analysis).
Consider the downstream consequences of a single missed STEMI:
- • The patient is transported to a facility without PCI capability, adding 30–90+ minutes of delay.
- • Critical reperfusion therapy is not initiated. Myocardial tissue dies at a rate that accelerates with every passing minute.
- • The patient develops heart failure, cardiogenic shock, or dies — from a condition that was treatable if identified on time.
- • A wrongful death lawsuit is filed. Industry estimates place the average indemnity payment for heart attack malpractice claims at approximately $400,000 — contributing to an estimated $2.4 billion in annual wrongful death exposure across the U.S. (based on published misdiagnosis and malpractice data).
Conditions like aortic dissection, intracerebral hemorrhage, and pulmonary thromboembolism can also cause ST-segment elevation. If anticoagulants are administered based on a false STEMI diagnosis triggered by electrode artifact, the consequences can be catastrophic — actively accelerating hemorrhage in a bleeding patient.
⚠ WARNING: A patient with an aortic dissection who receives heparin because an ECG artifact was misread as STEMI may bleed out internally. A patient with an intracerebral hemorrhage given anticoagulants faces a dramatically increased risk of death. These are not hypothetical scenarios. They are documented clinical consequences of misplaced electrodes leading to misdiagnosis.
A $2.4 Billion Problem That No One Is Talking About
Over 300 million ECGs are performed globally each year. With an electrode misapplication rate exceeding 90% by some measures, the scale of potentially compromised cardiac diagnostics is staggering. This is not a niche clinical concern. It is a systemic crisis affecting every emergency department, every ambulance, every ICU, and every cath lab in the developed world.
The financial bleeding is relentless:
- • False cath lab activations at rates of 15–40% burn through staff time, equipment, and medications while contributing to burnout and degraded emergency response quality.
- • Missed STEMIs result in extended ICU stays, heart failure management, long-term disability care, and wrongful death litigation — costs that can reach into the millions per patient.
- • Serial ECG discrepancies between prehospital and hospital recordings (caused by different electrode placements) create diagnostic confusion, trigger repeat testing, and delay definitive treatment.
- • Malpractice exposure from missed or delayed MI diagnoses represents an estimated $2.4 billion in annual wrongful death liability (1.5% misdiagnosis rate, 40% malpractice win rate, $400K average indemnity).
The SCAI expert consensus update on best practices in the cardiac catheterization laboratory — endorsed by the ACC, AHA, and HRS in 2021 — emphasized the importance of cost-per-case analysis and lab utilization optimization. Yet the single most addressable variable in this entire chain — the accuracy of ECG electrode placement at the point of acquisition — has received almost no technological innovation in seven decades.
What the Guidelines Demand — and Where the System Fails
The 2025 ACC/AHA/ACEP/NAEMSP/SCAI Guideline for the Management of Patients With Acute Coronary Syndromes is unequivocal: an ECG should be acquired and interpreted within 10 minutes of a patient presenting with symptoms suggestive of ACS. For STEMI, primary PCI should be performed within 90 minutes of first medical contact at a PCI-capable center, or within 120 minutes if transfer is required.
These timelines are aggressive by design. Research from the AHA’s Get With The Guidelines registry found that in-hospital STEMI mortality increased from 5.6% to 8.7% between 2018 and 2021, with only 17% of patients transferred from non-PCI-capable hospitals meeting the 120-minute treatment benchmark (JAMA, 2022).
The system is already failing to meet its own standards. Now layer on the reality that the ECG driving every critical decision in this chain may be corrupted from the moment the electrodes are placed.
The guidelines assume a valid ECG. The evidence proves we rarely have one.
The Path Forward: Technology Must Close the Gap That Training Cannot
Education and training help, but they are not enough. Research shows that more accurate electrode placement correlates with classroom-based training, frequent practice, and greater self-confidence (Clopton & Hyrkäs, 2024). But even among trained professionals who perform ECGs regularly, the error rates remain unacceptably high. Medani et al. (2018) demonstrated that peer-led educational interventions improved performance — but 90% of participants still could not place all leads correctly even after training.
The problem is not that clinicians are careless. The problem is that the technology demands anatomical precision under conditions that make precision nearly impossible: obese patients, female patients, patients drenched in sweat, patients in the back of a moving ambulance, patients in cardiac arrest. Individual adhesive electrodes applied one at a time, based on manual landmark identification, in a moment of extreme time pressure — this is a recipe for the failure rates we see.
C-Booth Innovations developed the EXG™ Wearable ECG Platform to fundamentally eliminate this vulnerability. By consolidating all electrode positions into a single, anatomically guided, wearable system, the EXG removes the guesswork from ECG acquisition. A clinician aligns the central portion of the device over the sternum with a nipple-line marker, and all six precordial electrodes are simultaneously placed in their correct anatomical positions.
The result: diagnostic-quality ECG data acquired in under 60 seconds, with dramatically reduced placement variability, from the ambulance to the emergency department to the cath lab — continuous, comparable monitoring across the entire episode of care.
The EXG platform is:
- • FDA registered and ISO certified
- • Designed to AHA-compliant standards
- • Expandable from 12-lead to 18-lead for enhanced STEMI detection
- • Radiolucent — compatible with CT, X-ray, and interventional imaging
- • Universally compatible with existing cardiac monitors
The Uncomfortable Truth
Every statistic in this article comes from a peer-reviewed study, a national registry, or a guideline endorsed by the American Heart Association, the American College of Cardiology, or the Heart Rhythm Society. None of this is speculative.
The uncomfortable truth is this: the most fundamental step in cardiac emergency diagnostics — placing ten electrodes on a patient’s body in the correct locations — is being done wrong the overwhelming majority of the time. And that failure is:
- • Killing patients whose heart attacks are masked by electrode artifact
- • Wasting billions in false cath lab activations, unnecessary angiography, extended hospitalizations, and malpractice settlements
- • Eroding trust as cath lab teams become desensitized to a flood of false alarms
- • Creating legal exposure of $2.4 billion annually in wrongful death claims from missed MIs
⚠ WARNING: Every ECG acquired with misplaced electrodes is a diagnostic gamble. Some patients win. Some patients lose everything. The question is: how long will we keep gambling with a 70-year-old technology when a better answer exists?
References
- Gregory P, Kilner T, Lodge S, Paget S. Accuracy of ECG chest electrode placements by paramedics: an observational study. British Paramedic Journal. 2021;6(1):8-14.
- Degheim G, Berry A, Zughaib M. False activation of the cardiac catheterization laboratory: The price to pay for shorter treatment delay. JRSM Cardiovascular Disease. 2019;8:2048004019836365.
- De Luca G, Suryapranata H, Ottervanger JP, et al. Time delay to treatment and mortality in primary angioplasty for acute myocardial infarction: every minute of delay counts. Circulation. 2004;109:1223-1225.
- McCabe JM, et al. False-Positive Cardiac Catheterization Laboratory Activation Among Patients With Suspected STEMI. Archives of Internal Medicine. 2012.
- Scholz KH, et al. Impact of treatment delay on mortality in STEMI patients (FITT-STEMI trial). European Heart Journal. 2018;39(13):1065-1074.
- Hadjiantoni A, et al. Is the correct anatomical placement of the ECG electrodes essential to diagnosis in the clinical setting: a systematic review. Cardiology and Cardiovascular Medicine. 2021;5(2):182-200.
- Clopton E, Hyrkäs EK. Assessing the Accuracy of ECG Chest Electrode Placement by EMS and Clinical Personnel. International Journal of Paramedicine. 2024;(6):29-47.
- Newman-Toker DE, et al. Diagnostic Errors in the Emergency Department: A Systematic Review. AHRQ Publication No. 22(23)-EHC043. 2022.
- Kania M, et al. The effect of precordial lead displacement on ECG morphology. Medical & Biological Engineering & Computing. 2014.
- Walsh B. Misplacing V1 and V2 can have clinical consequences. American Journal of Emergency Medicine. 2018;36(5):865-870.
- Bond RR, et al. The effects of electrode misplacement on clinicians’ interpretation of the standard 12-lead ECG. European Journal of Internal Medicine. 2012;23(7):610-615.
- Medani S, et al. Accuracy in precordial ECG lead placement: improving performance through a peer-led educational intervention. Journal of Electrocardiology. 2018;51:50-54.
- Rao S, O’Donoghue ML, Ruel M, et al. 2025 ACC/AHA/ACEP/NAEMSP/SCAI Guideline for the Management of Patients With Acute Coronary Syndromes. Circulation. 2025.
- Naidu SS, et al. SCAI expert consensus update on best practices in the cardiac catheterization laboratory (endorsed by ACC, AHA, and HRS). 2021.
- AHA Get With The Guidelines: Treatment Time and In-Hospital Mortality Among Patients With STEMI, 2018-2021. JAMA. 2022;328:2033-2040.
- Rajaganeshan R, et al. Accuracy of ECG lead placement among healthcare professionals. Heart. 2008.
About C-Booth Innovations
C-Booth Innovations is pioneering precision in cardiac monitoring with the EXG™ Wearable ECG Platform — founded by two emergency room physicians, inspired by the clinical need to improve the standard of patient care. The EXG system is FDA registered, ISO certified, and designed around AHA-compliant standards.
To learn more or schedule a demonstration, visit cboothinnovations.com or call (760) 800-2109.
© 2026 C-Booth Innovations. All rights reserved. | 5835 Avenida Encinas, Suite 118, Carlsbad, CA 92008
Ready to Improve ECG Accuracy?
See how the EXG platform can standardize electrode placement across your organization.


